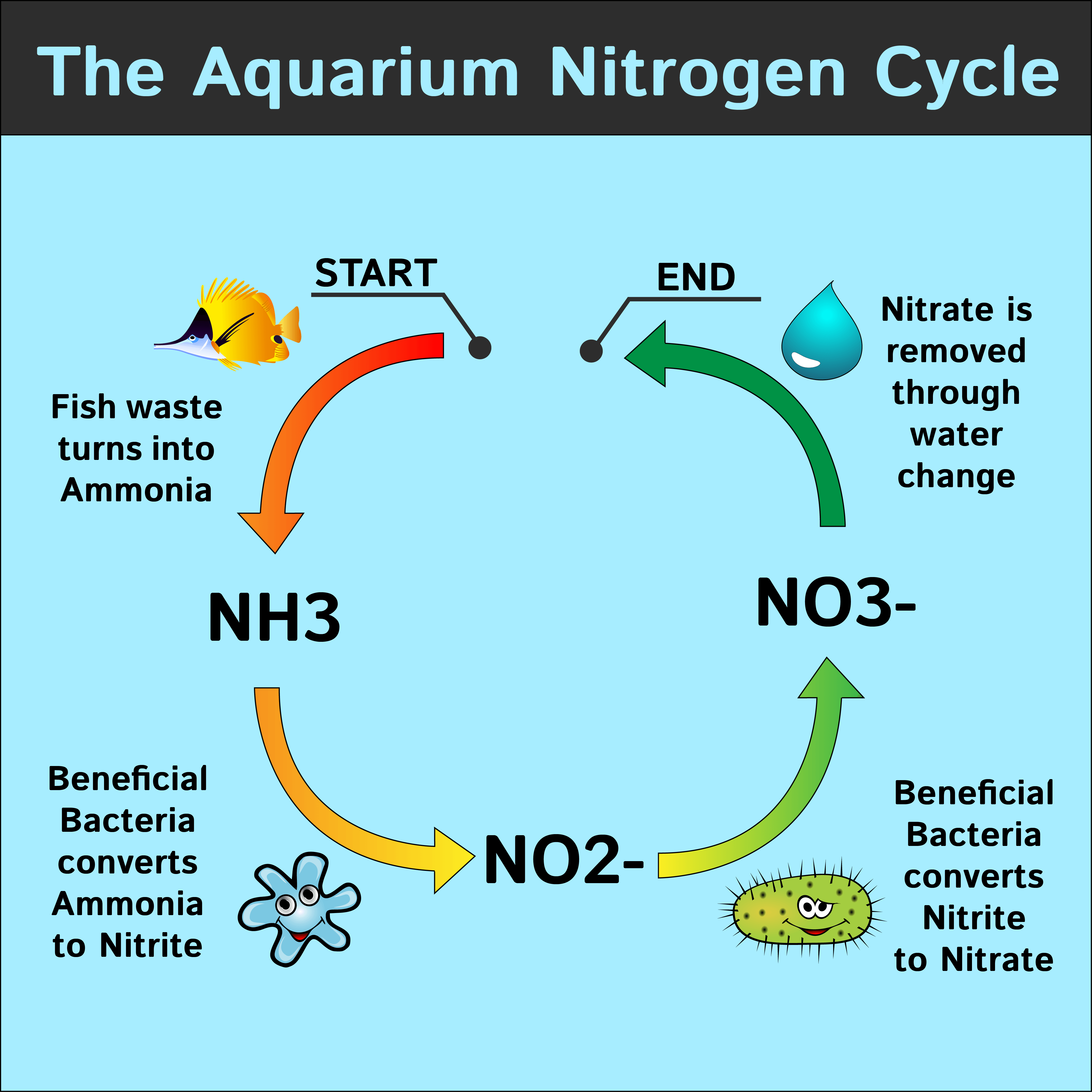
What is a “safe” level of ammonia for quarantine of fishes? | REEF2REEF Saltwater and Reef Aquarium Forum

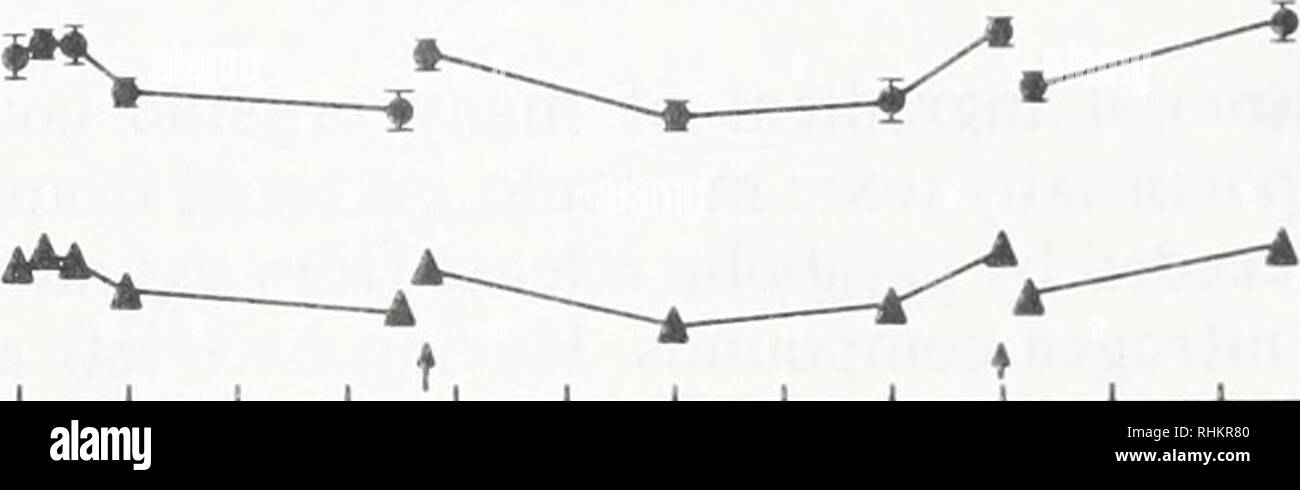
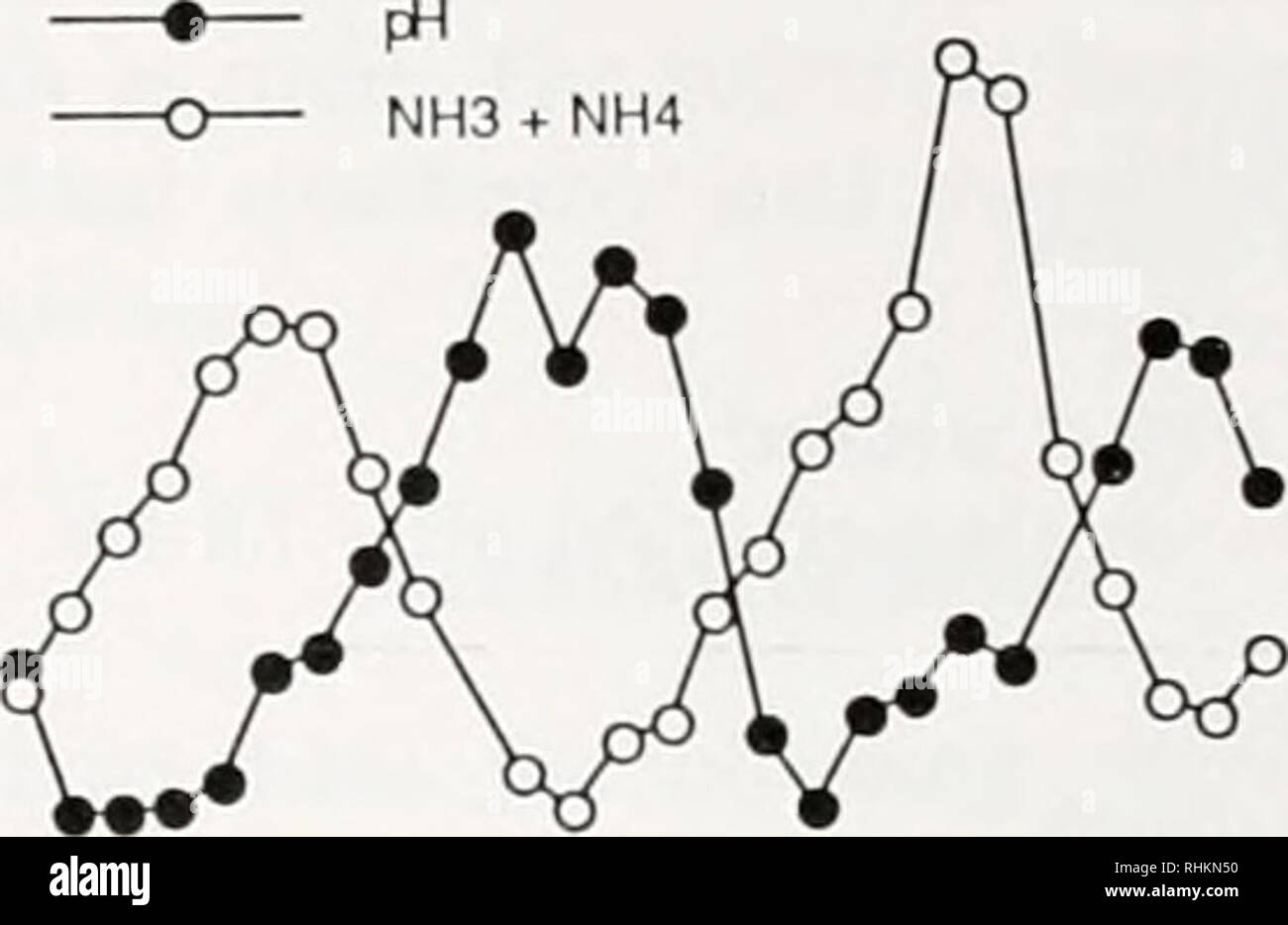
Rhesus Glycoprotein P2 (Rhp2) Is a Novel Member of the Rh Family of Ammonia Transporters Highly Expressed in Shark Kidney* - Journal of Biological Chemistry

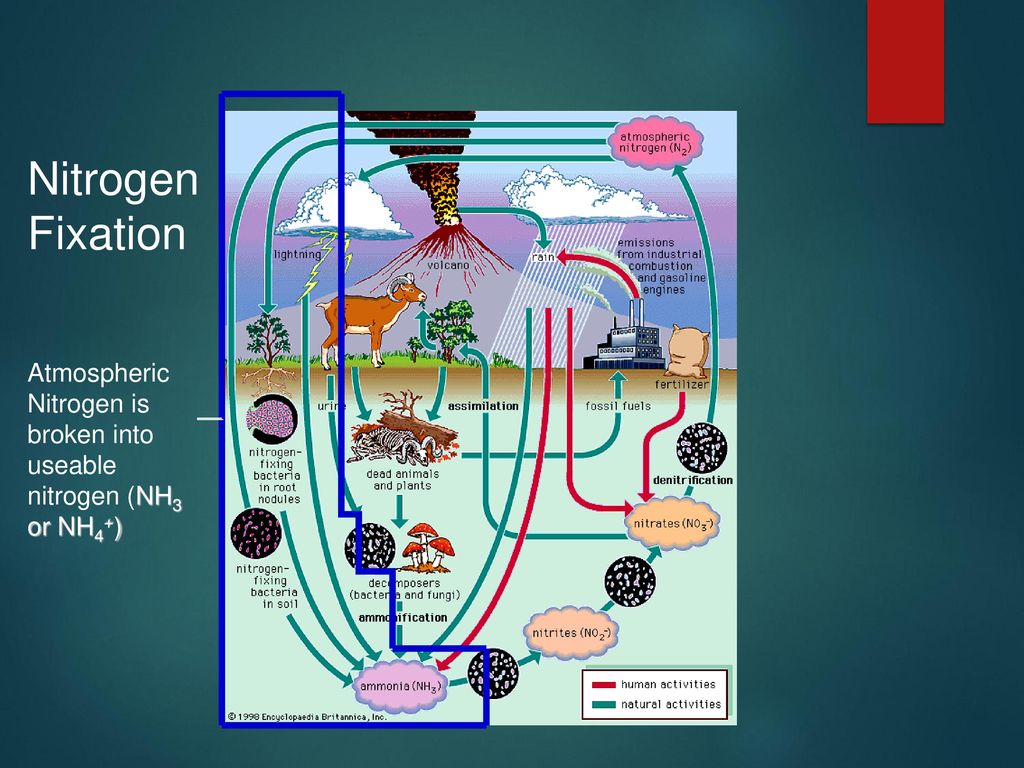
Rhcg1 and NHE3b are involved in ammonium-dependent sodium uptake by zebrafish larvae acclimated to low-sodium water | American Journal of Physiology-Regulatory, Integrative and Comparative Physiology

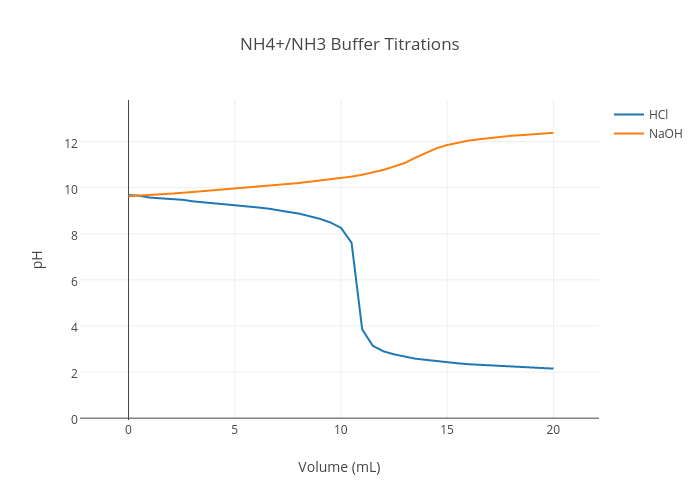
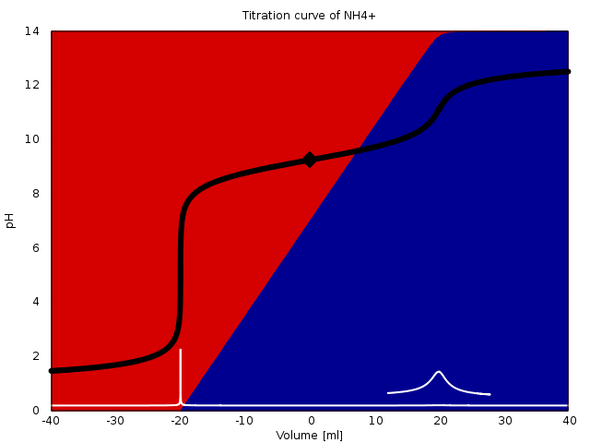
SOLVED:Calculate the pH change that results when 11 mL of 5.1 M NaOH is added to 764 mL of each the following solutions_ Use the Acid-Base Table (a) pure water (b) 0.10
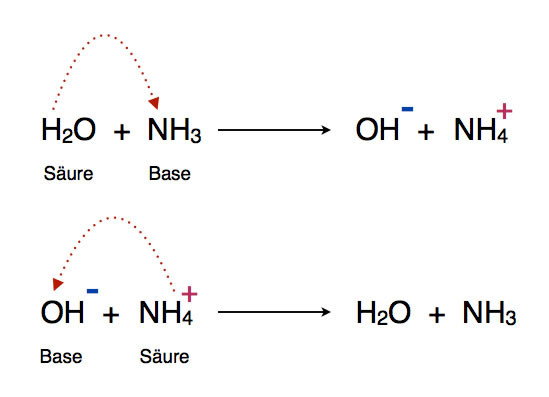
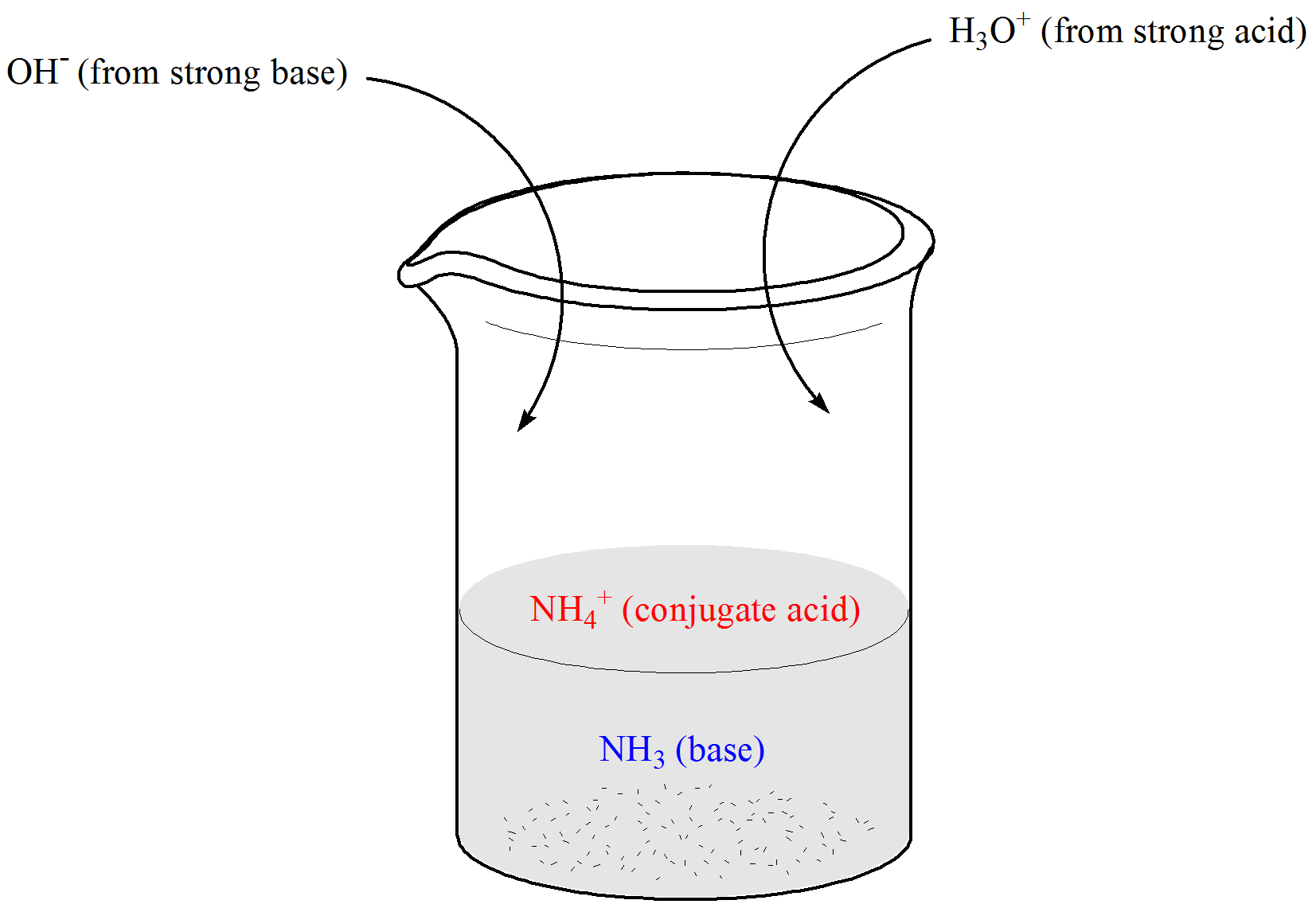
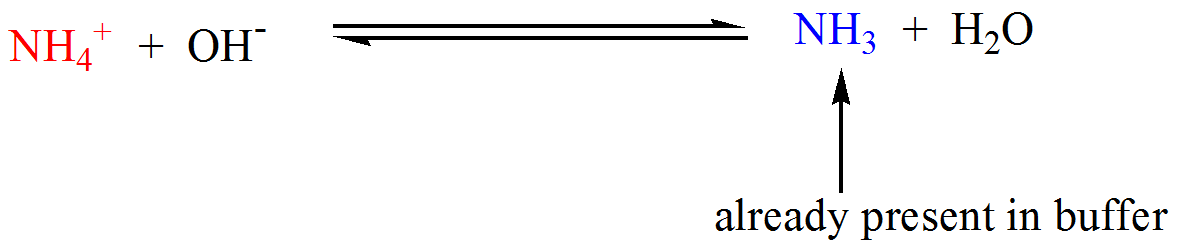
Welche Bedeutung hat die chemische Formel,,H2O+NH3- - - > OH- +NH4“ für die Säure-Base-Eigenschaften von Ammoniak? (Chemie, Säure-Base-Reaktion)